More unintended consequences
-
@Benjamin-Hall said in More unintended consequences:
Anything more? Nope. You dead.
That reminds me a bit of liquid bromine, which is technically less corrosive than HF but will merrily go through latex gloves and will do ungodly nasty things to body chemistry. I'm a software engineer for good reason…
-
@dkf said in More unintended consequences:
technically less corrosive
The best kind of less corrosive! Um...
-
-
@Jaloopa said in More unintended consequences:
@marczellm said in More unintended consequences:
What is a nfp?
Near Field Pornography
That's what happens when you video sexual activity close to but not in an agricultural area?
-
@dkf said in More unintended consequences:
liquid bromine
@HardwareGeek said in More unintended consequences:
@PleegWat Yes. Yes, it is. And both tetrafluoroammonium and phosphorus fluoride compounds are rather unpleasant. Related compounds PF5 and PF3 are described as "toxic" and "highly toxic," respectively. Tetrafluoroammonium likes to decompose into NF3 (which is surprisingly "safe" — it's only "immediately dangerous" at concentrations above 0.1%) and H2F+ ( ⇄ HF + H+ ), which does nasty things to living tissue.
@mott555 said in More unintended consequences:
@dkf Now I am become Death, the destroyer of worlds.
Looks like I posted the nope badger too soon.
SAVE YOURSELF!

-
@mott555 said in More unintended consequences:

This looks like something that would be featured in the chemistry equivalent of TDWTF.
-
@mott555 said in More unintended consequences:
@mott555 said in More unintended consequences:
@HardwareGeek said in More unintended consequences:
@dkf Upon further research, both NF4+ and PF6- are things that exist, so NF4PF6 is a plausible thing that could exist. It
would not be a pleasant thing to encounterseems unlikely that you would profit from an encounter with it.Okay, I'm finally excited for quantum computing. I'd love to play some kind of sandbox game that lets you create any chemical you want, such as this, and have it accurately simulate its effect on a sandbox world of some kind.
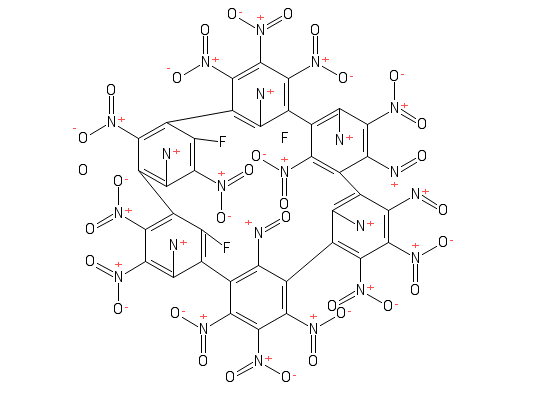
Fun with https://pubchem.ncbi.nlm.nih.gov/edit2/index.html
 Looks like you're trying to blow up, irradiate, and poison something.
Looks like you're trying to blow up, irradiate, and poison something.
-
@Zerosquare said in More unintended consequences:
This looks like something that would be featured in the chemistry equivalent of TDWTF.
It's got a lot of nitro groups, so it's going to be fun in the old explosive department anyway, but it the stochiometry combined with the stiffness of all those π-bonds (which will be very delocalised) which really scares me a lot; that's a molecule which just doesn't want to exist in this universe (for reasons related to thiotimoline). On the plus side, the likelihood that those fluorines will cause trouble afterwards is actually close to zero. I've no idea what they'll be bonded to, but bonded they certainly will.
-
@dkf said in More unintended consequences:
really scares me a lot; that's a molecule which just doesn't want to exist in this universe
This is basically what I was going for, since this was in the context of some kind of sandbox game that relies on quantum computing to accurately simulate chemistry on some level. Hack in a 55-gallon drum full of whatever you'd call that stuff, send your Sim into his house with it loaded on a hand truck, and then watch the fun begin when you unpause the chemical's simulation...
-
@dkf said in More unintended consequences:
@HardwareGeek said in More unintended consequences:
But it also does a pretty effective job of being very corrosive on direct contact.
IIRC, it's technically less corrosive than hydrochloric acid but way more “fun” in other ways. Like being happy to etch through glass and catastrophically decalcify your body fluids. I'm happy to never go anywhere close to the stuff…
I remember my dad saying that you can keep 85% HF(aq) in mild steel tanks, but 60% will eat straight through them.(1) He didn't mention why, but later education suggests that common ion effect probably has a role in it.
(1) Yes, it's more corrosive when you dilute it. I may be remembering the percentages wrongly (or he did, or both), of course, because they seem a bit on the high side. (Don't forget that I live in a country where you can buy 13% HCl(aq) in supermarkets.)
-
@Steve_The_Cynic said in More unintended consequences:
I remember my dad saying that you can keep 85% HF(aq) in mild steel tanks, but 60% will eat straight through them.
I think there's some sort of iron fluoride layer that forms reliably with the stronger solution.
-
@dkf said in More unintended consequences:
@Steve_The_Cynic said in More unintended consequences:
I remember my dad saying that you can keep 85% HF(aq) in mild steel tanks, but 60% will eat straight through them.
I think there's some sort of iron fluoride layer that forms reliably with the stronger solution.
Now that I think about it, that rings a bell. (And of course "common ion effect" ties in with the formation of that layer in high concentrations and its destruction in lower concentrations.)
-
@Gribnit said in More unintended consequences:
@mott555 said in More unintended consequences:
@mott555 said in More unintended consequences:
@HardwareGeek said in More unintended consequences:
@dkf Upon further research, both NF4+ and PF6- are things that exist, so NF4PF6 is a plausible thing that could exist. It
would not be a pleasant thing to encounterseems unlikely that you would profit from an encounter with it.Okay, I'm finally excited for quantum computing. I'd love to play some kind of sandbox game that lets you create any chemical you want, such as this, and have it accurately simulate its effect on a sandbox world of some kind.
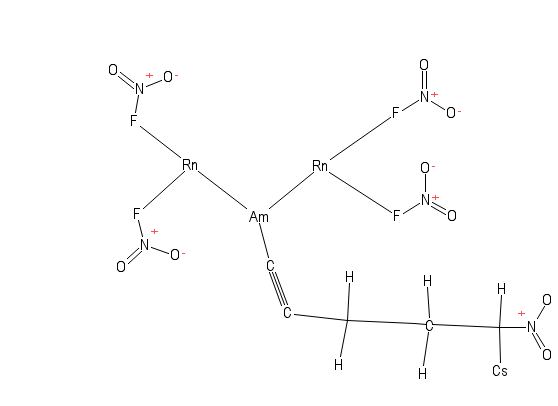
Fun with https://pubchem.ncbi.nlm.nih.gov/edit2/index.html

 Looks like you're trying to blow up, irradiate, and poison something.
Looks like you're trying to blow up, irradiate, and poison something.More like "blow up, irradiate and poison everything, all at once."
I'm pretty sure the caesium-carbon bond is going to give way in less than no time at all. And flourines with two bonds?
 That's gotta hurt.
That's gotta hurt.EDIT: Fluorines. Ugh. Spelling.
-
@Polygeekery said in More unintended consequences:
I don't want that much traffic to my tales.
Wait, you don't care about fake internet points and online fame? I don't understand what you're saying.
Also, is a story really a good story until you've heard at least 5 people calling it fake, been harassed about it via PM and had a moderator lock your post? I don't think so!
(JK, talesfromtechsupport is actually quite okay.)
-
@Polygeekery said in More unintended consequences:
I admit that one took me longer than it should have to find
Whenever somebody invents a foolproof system, God invents a bigger fool.
-
-
@dkf said in More unintended consequences:
@HardwareGeek said in More unintended consequences:
I believe that would be PNF in the system usually used for ionic/inorganic compounds
In the system for inorganic compounds, it'd depend on how they are bonded. But since F only ever makes one bond except under the most exotic of environments, the order NFP is… rather unexpected and the compound would be quite unstable.
Any time I hear chemists start using words like "exotic", "unexpected", or "unstable", I know that I don't want to be anywhere near whatever they're talking about.
-
@brie how about "interesting"?
-
@Gribnit When a physicist uses “interesting”, hold tight to your resonance cascades because you're possibly in for a wild ride…
-
@dkf said in More unintended consequences:
@Gribnit When a physicist uses “interesting”, hold tight to your resonance cascades because you're possibly in for a wild ride…
And make sure you have your trusty crowbar close at hand.
-
@Benjamin-Hall said in More unintended consequences:
@dkf said in More unintended consequences:
@Gribnit When a physicist uses “interesting”, hold tight to your resonance cascades because you're possibly in for a wild ride…
And make sure you have your trusty crowbar close at hand.
Not a problem. You'll surely be able to get at least a melee weapon early after the incident, and you can upgrade from any soldier corpses you come across.
-
So, I'm now the asshat who needs help.
I stuck my nose too far forward. I may even have cursed myself by using sentences like " how hard can it be?". Now I'm going be the admin of our network.
We've got a /27 from ISP1. I want automatic failover to isp2 (slow backup) but I want to fail back automatically as soon as ips 1 is up again.
We supply internet to a few others who currently have their own shitty customer grade routers in a shared switch directly from our ISP (one of many wtfs).
I want to have these after our router, preferably each on their own subnet. They must not be able to communicate with eachother, or our internal networks (employee and guest).
I need to have VPN access for all our users, but not anyone else. I'd like this to be fast-ish (100mbit full duplex)
I'd like to buy a few new APs, we currently have a single consumer router ( which is also a problem).
I'd like to use radius for VPN and WiFi. I need to rotate passwords for VPN 2-3 times a year, and if people quit.
The only 'real' equipment I've got is a ASA 5505 - currently unused. (And lots of dumb switches).
I'm considering the putting isps into the 5505, then into a vlan aware switch and a (untagged for external) port out to each of the networks fanned out from that. Then having 2-3 APs for our internal wlan networks . Our sub customers would probably be fine with a natted connection.
We're 20 users with 1-2 devices each who needs WiFi, current state is obviously bad.
I'd like a captive portal for our guest network.
I'm a ccna guy, so I'm considering buying Cisco. But I have no clue which gear is current, or if I can even get software updates.
If I pressure my boss I can probably get a budget of 2-3k USD.
Any recommendations? Both software and hardware are welcome. Hiring HPCs is off the table - we got screwed the last time (hence the 5505).
I'd like a solution I can scale up if more people are hired/ more people want to share our internet. I'd also like to be able to limit each customers bandwidth.
Did I mention downtime is frowned upon?
-
-
@swayde
Budget wise, you’re probably looking at a single NetGear or similar smart switch (48 port, ~$300), and then Meraki MR33s for the wireless.The ASA can be configured to fail over and fail back, you need to look into static route tracking
Assuming you have a Windows Active Directory for computer & server authentication, you can configure the ASA to use that via LDAP for per user vpn authentication and relieve the need to rotate passwords there. And you can use Azure Traffic Manager (or similar services from AWS or GCP) to publish public IPs on both providers so you get automatic failover on the vpn as well.
-
@pie_flavor said in More unintended consequences:
That was uncalled for. My sound was muted, but others might not be so lucky.
-
@marczellm said in More unintended consequences:
What is a nfp?
Near-field penis. A common angle for fellatio videos.
Edit: technical
 but mine is more specific.
but mine is more specific.
-
@izzion thanks for the suggestions, I'll look into it.
-
Unintended consequences: what happens when you speed-read, and think @swayde's post is a reply to @Tsaukpaetra's post just above.
-
@Gribnit said in More unintended consequences:
@brie how about "interesting"?
I don't think you'd necessarily need to start running like you would if they said "exotic", "unexpected", or "unstable".
@dkf said in More unintended consequences:
@Gribnit When a physicist uses “interesting”, hold tight to your resonance cascades because you're possibly in for a wild ride…
The nice thing about physicists is that they tend to either be working with things that are mainly theoretical, or else that are far too tiny to affect you directly in any significant way unless there was a tremendously huge number of them. And there is far less likely to be a huge number of those tiny things when you're around physicists than there is when you're around chemists.
When either a physicist or a chemist says "interesting" you should probably pay attention because it probably is pretty dang interesting (if only you can understand half of it).
-
@brie said in More unintended consequences:
When either a physicist or a chemist says "interesting" you should probably pay attention because it probably is pretty dang interesting (if only you can understand half of it).
Either that or you should be edging towards the door to avoid any pieces of lab equipment being lodged in you as shrapnel when the experiment explodes.
-
@e4tmyl33t said in More unintended consequences:
Either that or you should be edging towards the door to avoid any pieces of lab equipment being lodged in you as shrapnel when the experiment explodes.
It's not always exploding! Sometimes it is poisoning or irradiating or zapping or asphyxiating or…