The Official Status Thread
-
Status: I just read a brillant article that includes a warning that polystyrene contains a chemical known as styrene.
-
@Zenith said in The Official Status Thread:
@Zerosquare said in The Official Status Thread:
Why do you need a hardened laptop? Is it because you're expecting to bang your head against the keyboard frequently?

Because just about every consumer and business laptop that I've used aside from real IBM Thinkpads has been shit?
Putting a cruddy laptop in a big case doesn't change the rest of the laptop.
-
@Gąska said in The Official Status Thread:
Status: I just read a brillant article that includes a warning that polystyrene contains a chemical known as styrene.
“contains”
 !!! WARNING !!!
!!! WARNING !!! 
Sodium chloride contains the toxic gas chlorine! DO NOT EAT!
-
@kazitor it's worse than that. Polystyrene is nothing more, nothing less than a chain of styrene molecules (hence the name - literally "many styrenes").
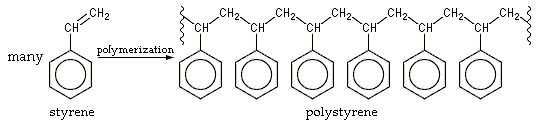
-
@HardwareGeek said in The Official Status Thread:
@topspin said in The Official Status Thread:
maybe it's a fucking nightmare.
Advice: Stay out of @Tsaukpaetra's dreams.
Get into his car!
Wait, maybe not.

-
@Tsaukpaetra said in The Official Status Thread:
@Gąska said in The Official Status Thread:
@Tsaukpaetra IT WAS SUPPOSED TO BE A JOKE GODDAMMIT!!!
It's OK. My uptime has now been reset to a sensible number again...
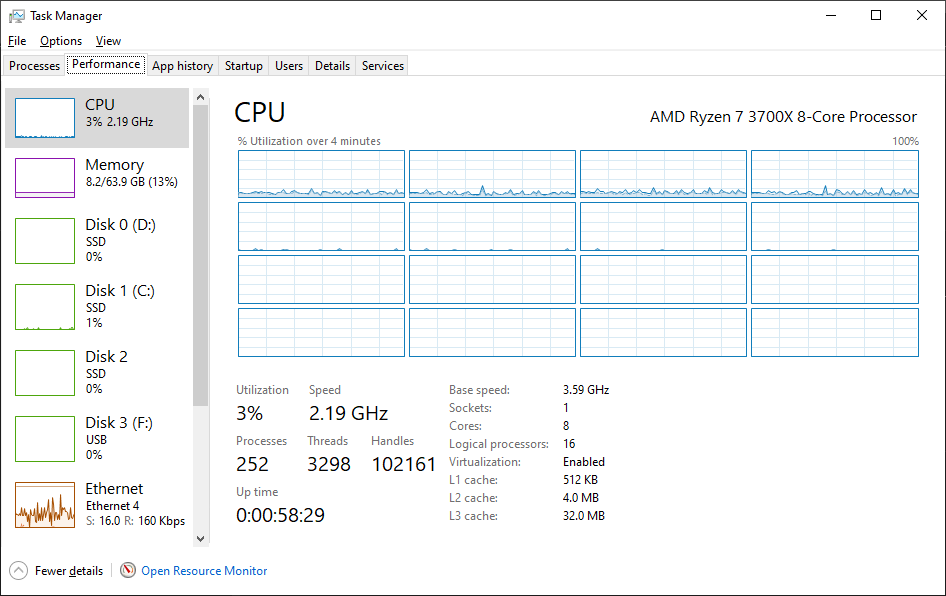
Likely an overflow issue. Mine will take some more time to reach 0 again:
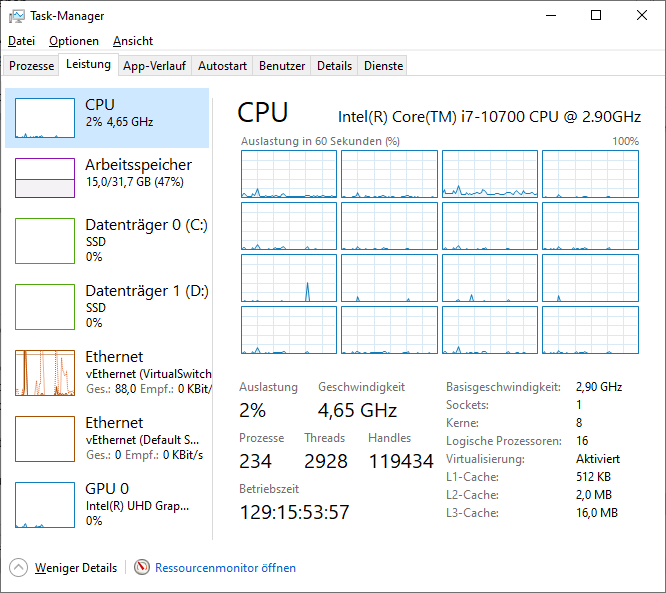
-
@Gąska said in The Official Status Thread:
@kazitor it's worse than that. Polystyrene is nothing more, nothing less than a chain of styrene molecules (hence the name - literally "many styrenes").

Oh guess... Sometimes, a lonely styrene molecule may be forgotten and remain lonely among those polys.
So, it's correct: there may be some monomeric styrene molecules left.
-
@topspin said in The Official Status Thread:
Fuck you "Verified by VISA", I hate you so. fucking. much.
This shit never works.That definitely depends at least in part on the bank. I've never had that problem with my (VISA) credit card.
-
@dkf said in The Official Status Thread:
@topspin said in The Official Status Thread:
Fuck you "Verified by VISA", I hate you so. fucking. much.
This shit never works.That definitely depends at least in part on the bank. I've never had that problem with my (VISA) credit card.
Well, this shitty app is definitely the bank's fault. But I've had it not-work back then, before it was using the bank's app and still redirected you to several insecure, shady looking sites run by Visa.
The card itself also works just fine in the 95% of times it doesn't request this garbage.
-
@dkf said in The Official Status Thread:
That definitely depends at least in part on the bank. I've never had that problem with my (VISA) credit card.
Nah, seems to mostly work. I've seen a few different implementations, on account of relocations. The most recent iteration seems to be somewhat useless (i.e., I don't have to do anything, it just sits at the verified-screen for a bit and then goes on its merry way). So far, I'm not complaining.
The one person that seems to struggle with it is my dad. But he recently retired and for some reason that has turned him into a technologically illiterate curmudgeon. So, I doubt the problem is with the system...
-
@cvi said in The Official Status Thread:
Nah, seems to mostly work. I've seen a few different implementations, on account of relocations. The most recent iteration seems to be somewhat useless (i.e., I don't have to do anything, it just sits at the verified-screen for a bit and then goes on its merry way). So far, I'm not complaining.
It might be that they're detecting somehow that you're using the same device that you used previously. Or they might just be half-assing it.

-
@dkf said in The Official Status Thread:
It might be that they're detecting somehow that you're using the same device that you used previously.
I'd hope it's this.
Or they might just be half-assing it.
But, realistically speaking, it's probably this.
-
@Gąska said in The Official Status Thread:
Status: I just read a brillant article that includes a warning that polystyrene contains a chemical known as styrene.
Next you're gonna tell us that @Polygeekery contains geekery.
-
Status: I just looked at the Jenkins server and realized, with slight horror, that its "authorization strategy" is set to "anyone can do anything".

How did that happen? What fucking update broke this??
I just clicked "matrix-based security", which is the way it used to be, and the user matrix I had set up is also completely gone. There's just the groups "anonymous users" and "authenticated users". So let's just enable nothing / everything respectively first before I recreate the permissions matrix.And no, I definitely did not dream this up, there's a backup config file lying around there that still contains the old permissions.
-
@topspin said in The Official Status Thread:
I just clicked "matrix-based security", which is the way it used to be, and the user matrix I had set up is also completely gone.
Yeah. Jenkins is extremely retarded in that, if you disable anything, it immediately deletes any possible configuration you might have had for that module.
Guess how I found out when I was fiddling with the authentication server settings?
-
@Tsaukpaetra said in The Official Status Thread:
@topspin said in The Official Status Thread:
I just clicked "matrix-based security", which is the way it used to be, and the user matrix I had set up is also completely gone.
Yeah. Jenkins is extremely retarded in that, if you disable anything, it immediately deletes any possible configuration you might have had for that module.
Guess how I found out when I was fiddling with the authentication server settings?
Thankfully, for me there's no real attack scenario here. Of course you could damage a lot of things if you mess with this, but authentication or not, anyone who can even reach the server address has enough access to potentially create worse problems, anyway.
As Sutter used to say, I'm just guarding against Murphy, not Machiavelli.That's also why I'm getting annoyed of their weekly "install teh updatez now!!1!".
-
@Gąska said in The Official Status Thread:
@kazitor it's worse than that. Polystyrene is nothing more, nothing less than a chain of styrene molecules (hence the name - literally "many styrenes").

Table salt is nothing more, nothing less than a matrix of flammable metal and toxic gas.
-
-
@kazitor said in The Official Status Thread:
Table salt is nothing more, nothing less than a matrix of flammable metal and toxic gas.
They're both ionized. Oh no!

-
-
@cvi, sincerest apologies.
Clearly I should have written “inflammable.”

-
@Zenith Would anybody be surprised to hear that I was tired of waiting for support to figure out what they broke and fixed it myself by changing a config file that hasn't been touched since 2014?
-
@cvi said in The Official Status Thread:
he recently retired and for some reason that has turned him into a technologically illiterate curmudgeon.
I still have a few years until retirement, but I've already got a good start on the curmudgeon part.

-
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
@kazitor it's worse than that. Polystyrene is nothing more, nothing less than a chain of styrene molecules (hence the name - literally "many styrenes").

Table salt is nothing more, nothing less than a matrix of flammable metal and toxic gas.
This is technically incorrect but I realize you're just going for a cheap joke so I'll spare you the explanation.
-
-
@Zerosquare well, saturated geekery.
-
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
This is technically incorrect
Now do polymers!
Doing inanimate substances sounds more like a job for @Tsaukpaetra.

-
@HardwareGeek said in The Official Status Thread:
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
This is technically incorrect
Now do polymers!
Doing inanimate substances sounds more like a job for @Tsaukpaetra.

At least they are organic!
-
Status: Can't ever seem to get ahead of myself. I moved into this place with half of my worldly possessions using about a dozen storage totes. I must be approaching 250 and I still can't see more than bits and pieces of the floor. What the fuck happened?
-
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
This is technically incorrect
Now do polymers!
Serious question: which part did I get wrong? I was never very good at chemistry but I'm pretty sure I've got everything right there. Maybe except the word "molecule" but is there a better word for something that used to be a separate molecule and now is part of a polymer?
-
@Gąska said in The Official Status Thread:
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
This is technically incorrect
Now do polymers!
Serious question: which part did I get wrong? I was never very good at chemistry but I'm pretty sure I've got everything right there. Maybe except the word "molecule" but is there a better word for something that used to be a separate molecule and now is part of a polymer?
I assume the part where the chemical properties of polymers are different from a soup of monomers, just like (but maybe less pronounced) the properties of salt are different from those of sodium and chlorine.
-
Status: New PC build crashed hard last night in the most damning way possible - "no signal" with only the power LED remaining on. I'm so not looking forward to pissing away days on end to find the cause

-
@Applied-Mediocrity I assume therefore is wasn't the obvious "bad display cable".
-
@topspin said in The Official Status Thread:
@Gąska said in The Official Status Thread:
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
This is technically incorrect
Now do polymers!
Serious question: which part did I get wrong? I was never very good at chemistry but I'm pretty sure I've got everything right there. Maybe except the word "molecule" but is there a better word for something that used to be a separate molecule and now is part of a polymer?
I assume the part where the chemical properties of polymers are different from a soup of monomers, just like (but maybe less pronounced) the properties of salt are different from those of sodium and chlorine.
Damn, I could swear I never said that. But just in case I did, thanks for correction.
(Just in case it wasn't clear - I seriously can't tell how much it's @kazitor trolling me and how much it's being even worse at chemistry than me - the difference between polymers and chloride salts is that creating salts from chlorine gas requires splitting chlorine molecules in half, while no such thing happens during polymerization.)
-
Status: Every TVTropes page I visited today seems to have a few entries that made me think "the person who wrote this doesn't have any idea what this trope is supposed to be about". Kinda ruins the fun.
-
@BernieTheBernie said in The Official Status Thread:
@Gąska said in The Official Status Thread:
@kazitor it's worse than that. Polystyrene is nothing more, nothing less than a chain of styrene molecules (hence the name - literally "many styrenes").

Oh guess... Sometimes, a lonely styrene molecule may be forgotten and remain lonely among those polys.
So, it's correct: there may be some monomeric styrene molecules left.Only if the person in charge of building the molecule failed to tighten all the nuts correctly.
-
@Gąska said in The Official Status Thread:
Serious question: which part did I get wrong?
Part one: “Polystyrene is nothing more, nothing less than a chain of styrene molecules.” Part two: mixing condescension with untruth.
“chain of styrene,” fine. “Nothing more, nothing less” = literally the molecules, which it isn’t. Styrene’s as liquid as petrol, for one, whereas PS is as liquid as the American name for petrol.
@Gąska said in The Official Status Thread:
creating salts from chlorine gas requires splitting chlorine molecules in half, while no such thing happens during polymerization.
Splitting double bonds “in half” doesn’t count, evidently. You posted the image; you could have at least looked at it.
-
@kazitor said in The Official Status Thread:
@Gąska said in The Official Status Thread:
Serious question: which part did I get wrong?
Part one: “Polystyrene is nothing more, nothing less than a chain of styrene molecules.” Part two: mixing condescension with untruth.
“chain of styrene,” fine. “Nothing more, nothing less” = literally the molecules, which it isn’t.
I'll give you that. I meant from a structural viewpoint, but I see how it could be understood differently.
@Gąska said in The Official Status Thread:
creating salts from chlorine gas requires splitting chlorine molecules in half, while no such thing happens during polymerization.
Splitting double bonds “in half” doesn’t count, evidently.
Should it? I seriously don't know how polymerization really works, on a nanosecond-by-nanosecond timescale, and how significant it is that for a very brief moment the double bonds are replaced by single bonds in order to later combine with other styrene molecules. To my simple uneducated mind, this seems much less notable than blowing up full molecules into single atoms.
You posted the image; you could have at least looked at it.
I did. Everything I wrote above still applies. The particles of the "toxic gas" you wrote about aren't contained in particles of sodium chloride. The particles of styrene are contained in full in polystyrene.
And no, @Tsaukpaetra, I won't split it into separate topic. If you're that much bothered by it, you know where the report button is.
-
@Gąska said in The Official Status Thread:
The particles of the "toxic gas" you wrote about aren't contained in particles of sodium chloride. The particles of styrene are contained in full in polystyrene.
In either case, the new molecules still contain the atoms of the old molecules. Which isn't particularly relevant. In both cases, the chemical properties of the starting material and product are quite different.
-
@topspin it is relevant in context of "polystyrene getting into your food is dangerous because it dissolves into styrene, which is toxic". No such risk (as far as I know) exists with NaCl, specifically because it's not structurally made of Cl2, so instead it dissolves into Cl- and that for some reason isn't nearly as toxic as Cl2. I may have gotten some facts wrong here, though.
-
@Zecc My own builds are always cursed with something similar to The Curse of @Tsaukpaetra. Except this time it's been working for some time now. I ceased being vigilant and that's when it decided to strike.
-
@Gąska said in The Official Status Thread:
And no, @Tsaukpaetra, I won't split it into separate topic. If you're that much bothered by it, you know where the report button is.
I am shocked and dismayed.
-
@Gąska said in The Official Status Thread:
@topspin it is relevant in context of "polystyrene getting into your food is dangerous because it dissolves into styrene, which is toxic".
That may be so, and probably what you had in mind. But your post read like "basically is a bunch of styrene" and not "dissolves into styrene".
-
@topspin said in The Official Status Thread:
@Gąska said in The Official Status Thread:
@topspin it is relevant in context of "polystyrene getting into your food is dangerous because it dissolves into styrene, which is toxic".
That may be so, and probably what you had in mind. But your post read like "basically is a bunch of styrene" and not "dissolves into styrene".
Except I didn't say "basically is a bunch of styrene". I said something completely different which may sound if you're just skimming through and have enough bad faith like I said that. I still stand by my claim that @kazitor's statement "there's toxic gas inside kitchen salt" is orders of magnitude more wrong than my statement "there's styrene inside polystyrene". I may be mistaken about it and there might be legit reason why thinking of polystyrene as "styrene molecules chained together into a longer molecule" is fundamentally wrong. But "chemical properties of the starting material and product are quite different" isn't that. Of course they are different. They're different molecules, why wouldn't they be different. But the fact that polystyrene easily dissolves into a bunch of styrene, while sodium chloride doesn't dissolve into chlorine gas, suggests I'm more right than wrong.
Just to reiterate. This whole argument started with @kazitor indirectly pointing out that I'm wrong to say that polystyrene contains styrene. I'm trying to figure out whether I'm actually wrong or not. You and your simplistic points about different molecules having different properties don't help.
-
@Tsaukpaetra said in The Official Status Thread:
@Gribnit said in The Official Status Thread:
@HardwareGeek said in The Official Status Thread:
@topspin said in The Official Status Thread:
maybe it's a fucking nightmare.
Advice: Stay out of @Tsaukpaetra's dreams.
Hey! No interpretation!
People can be in my dreams, no need for interpretation there!
Stop that. There's kids reading.
-
@Gąska said in The Official Status Thread:
The particles of styrene are contained in full in polystyrene.
True for polystyrene, but not for all polymers. Some polymerise via condensation reactions, where a molecule of water is removed for every connection made between monomer units. This is a particularly important process in biological polymers (both proteins and complex sugars).
-
@Gąska said in The Official Status Thread:
instead it dissolves into Cl- and that for some reason isn't nearly as toxic as Cl2
Chloride ions are far more stable than chlorine molecules (due to having the same electronic configuration as argon) despite the charge imbalance. That's true for all the halogens at least down to iodine (things get radioactive and screwy below that) and any toxicity is usually just due to solubility of their salts being such that they remove some important cations from the blood (such as calcium with fluoride) or their physical size causing problems in ion channels.
You have a lot of chloride in your blood; it's one of the most common anions (along with bicarbonate).
-
@kazitor said in The Official Status Thread:
Splitting double bonds “in half” doesn’t count, evidently. You posted the image; you could have at least looked at it.
Aaagh.
Stop it, just stop it, none of this is how it works. A double bond doesn't get split, it ends up acting as an electron donor but the covalent bond is not broken, reaction energies would be much higher.
-
@Gąska said in The Official Status Thread:
This whole argument started with @kazitor indirectly pointing out that I'm wrong to say that polystyrene contains styrene. I'm trying to figure out whether I'm actually wrong or not.
You're right by @BernieTheBernie's observation regarding unreacted monomer. But, the chemical specie that is a single monomer unit in a polystyrene chain is much less reactive than a free styrene monomer.
-
